CAS No.: 1226781-44-7
奥格列汀 Catalog No. CSN13845
Synonyms: MK-3102
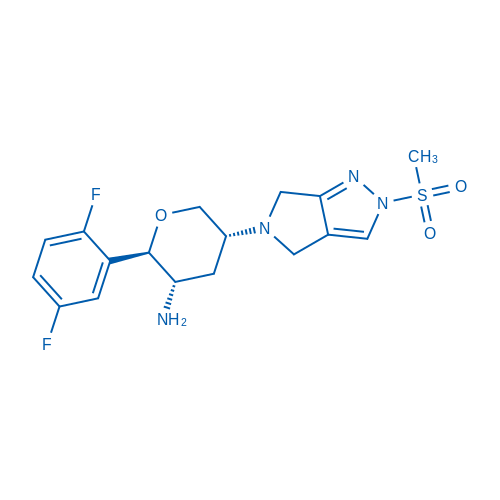
Omarigliptin, also called MA-3102, is an selective inhibitor of DPP-4 with IC50 of 1.6 nM and Ki of 0.8 nM. Its selectivity is higher than other 168 proteasomes.
| 规格 | 价格 | 促销价格 | 库存 | 数量 |
|---|

纯度 & 质量文件
批次:
靶点选择性
生物活性
靶点 DPP4
IC50:1.6nM- 描述
- 临床研究
NCT号 适应症或疾病 临床期 招募状态 预计完成时间 地点 NCT01217073 Type 2 Diabetes Mellitus Phase 2 Completed - - NCT01682759 Type 2 Diabetes Mellitus Phase 3 Completed - - NCT01841697 Type 2 Diabetes Phase 3 Completed - - - 更多
- 参考文献
- [1] Sheu WH, Gantz I, et al. Safety and Efficacy of Omarigliptin (MK-3102), a Novel Once-Weekly DPP-4 Inhibitor for the Treatment of Patients With Type 2 Diabetes. Diabetes Care. 2015 Nov;38(11):2106-14.
- [2] Biftu T, Sinha-Roy R, et al. Omarigliptin (MK-3102): a novel long-acting DPP-4 inhibitor for once-weekly treatment of type 2 diabetes. J Med Chem. 2014 Apr 24;57(8):3205-12.
- [3] Biftu T, Sinha-Roy R, Chen P, Qian X, Feng D, Kuethe JT, Scapin G, Gao YD, Yan Y, Krueger D, Bak A, Eiermann G, He J, Cox J, Hicks J, Lyons K, He H, Salituro G, Tong S, Patel S, Doss G, Petrov A, Wu J, Xu SS, Sewall C, Zhang X, Zhang B, Thornberry NA, Weber AE. Omarigliptin (MK-3102): a novel long-acting DPP-4 inhibitor for once-weekly treatment of type 2 diabetes. J Med Chem. 2014 Apr 24;57(8):3205-12. doi: 10.1021/jm401992e. Epub 2014 Apr 2. PMID: 24660890.
- [4] Tan X. Omarigliptin for the treatment of type 2 diabetes. Endocrine. 2016 Oct;54(1):24-31. doi: 10.1007/s12020-016-1011-9. Epub 2016 Jul 2. PMID: 27372109.
- [5] Krishna R, Addy C, Tatosian D, Glasgow XS, Gendrano Iii IN, Robberechts M, Haazen W, de Hoon JN, Depré M, Martucci A, Peng JZ, Johnson-Levonas AO, Wagner JA, Stoch SA. Pharmacokinetics and Pharmacodynamics of Omarigliptin, a Once-Weekly Dipeptidyl Peptidase-4 (DPP-4) Inhibitor, After Single and Multiple Doses in Healthy Subjects. J Clin Pharmacol. 2016 Dec;56(12):1528-1537. doi: 10.1002/jcph.773. Epub 2016 Jun 17. PMID: 27225334; PMCID: PMC5111764.
实验方案
- 计算器
- 储存液制备
技术信息
| CAS号 | 1226781-44-7 | 储存条件 |
|
|||||||
| 分子式 | C17H20F2N4O3S | 运输 | 蓝冰 | |||||||
| 分子量 | 398.43 | 别名 | MK-3102 | |||||||
| 溶解度 |
|
动物实验配方 |
 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
